For decades, HIV drugs focused on viral enzymes. Lenacapavir changed that by targeting the virus’s structural core. This article traces how the HIV capsid, once thought to be a passive shell, emerged as a dynamic and essential driver of infection, and how structure-guided research turned it into a powerful drug target. From capsid architecture and host protein interactions to the development of the first long-acting capsid inhibitor, this piece explains how lenacapavir disrupts HIV at multiple stages of its life cycle and what this breakthrough means for treatment, prevention, and future antiviral design.
The HIV Capsid: The Path to Lenacapavir
HIV infection begins when the virus binds to receptors on an immune cell, triggering fusion between the viral envelope and the cell membrane, releasing the intact capsid (CA) protein and viral genome into the cytoplasm. Since the mid-2000s, the capsid has been recognized as a dynamic structure that interacts with more than 20 human host proteins such as CPSF6 and Nup153 to help the virus navigate through the cell and reach the nucleus. This capsid lattice comprising hexamers and pentamers is a striking example of evolutionary design: it must be stable enough to protect the genome during transport yet flexible enough to disassemble at the nuclear pore so the genome can enter the nucleus. Because the capsid is essential for reverse transcription and nuclear entry, it is a powerful, though challenging target for antiviral drug development.
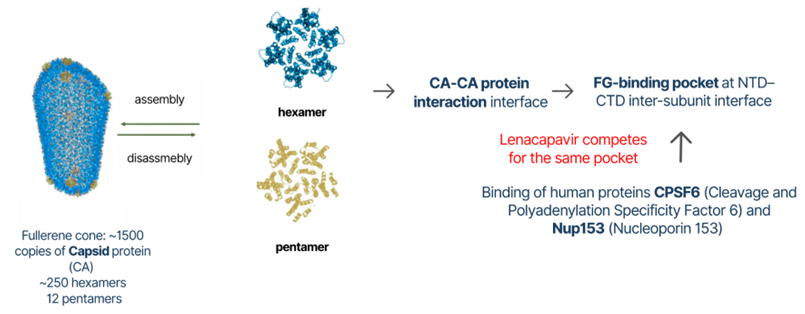
There are currently 49 FDA-approved HIV medicines that target different stages of the viral life cycle, from entry and fusion to viral enzymes such as reverse transcriptase, protease, and integrase. A key shift came in 2010 with PF-74, which showed that capsid protein interfaces could be targeted by drugs, even though PF-74 itself was not suitable for clinical use because it was rapidly broken down. Building on these structural insights, Gilead Sciences refined the chemistry to improve potency and stability, ultimately leading to the development of lenacapavir (GS-6207).
Lenacapavir: Turning Structure into Therapy
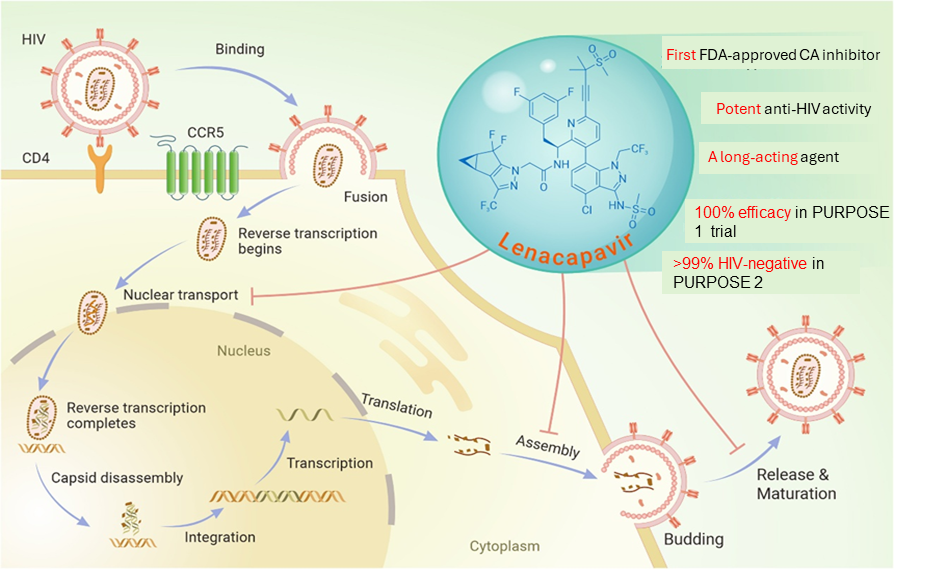
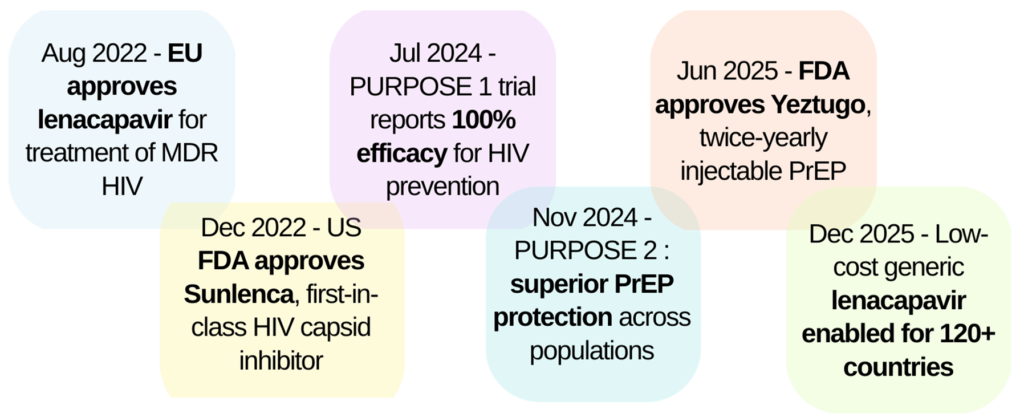
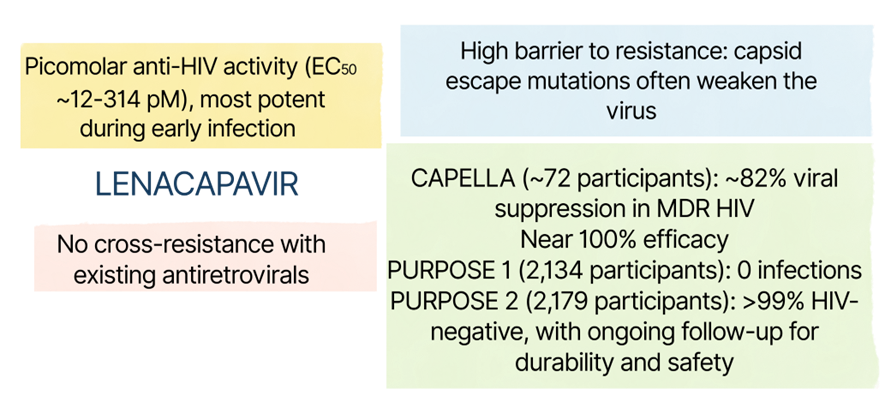
Lenacapavir is the first approved drug to directly target the HIV-1 capsid. Developed over nearly 15 years of research, lenacapavir (GS-6207, Sunlenca®, Yeztugo®) represents a new class of antivirals that work by targeting the virus’s structure rather than its enzymes. By binding a highly conserved region of the capsid, lenacapavir is effective across different HIV types and against many drug-resistant strains.
Lenacapavir works in two key phases of the HIV life cycle. Early on, it over-stabilizes the capsid, a process known as lethal hyperstabilization, which locks the capsid subunits together and removes the flexibility needed for uncoating, reverse transcription, and host-guided nuclear entry. Later, during virus assembly, lenacapavir disrupts capsid geometry by favoring flat hexamers and blocking pentamer formation, leading to malformed, non-infectious virus particles. Interestingly, capsid mutations that allow escape from lenacapavir often weaken the virus itself, creating a high genetic barrier to resistance.
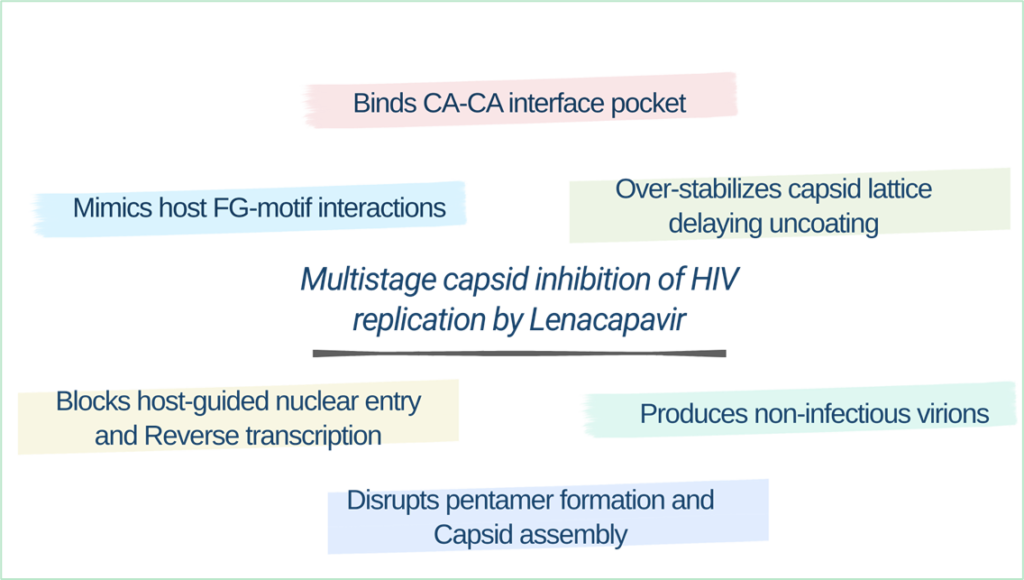
Lenacapavir has shown impressive results in multiple clinical trials for both HIV treatment and prevention, supported by its long half-life and twice-yearly injection schedule. However, real-world data, especially from low-resource settings, are still ongoing. Long-term safety is being closely monitored including possible effects on the brain. Few resistant mutations reported so far has usually been linked to poor adherence to other HIV medicines, highlighting why lenacapavir must be used as part of combination therapy.
Altogether, these advances point to a new way of thinking about antiviral drug design. Lenacapavir shows that viral structures once thought impossible to drug can be powerful targets. By understanding the 3D architecture of the HIV capsid, researchers designed a drug that disrupts the virus’s structure itself, not just a single enzyme step.
Further read
- https://www.gilead.com/news/news-details/2025/yeztugo-lenacapavir-is-now-the-first-and-only-fda-approved-hiv-prevention-option-offering-6-months-of-protection
- Rossi, E., Meuser, M. E., Cunanan, C. J., & Cocklin, S. Structure, function, and interactions of the HIV-1 capsid protein. Life (Basel). 2021;11(2):100. https://doi.org/10.3390/life11020100
- Hui, Z., Ye, X.-Y., Zhang, Y., et al. Lenacapavir, a first-in-class, long-acting capsid inhibitor approved for both HIV-1 treatment and prevention. Medicinal Chemistry Research. 2025;34:2472–2485. https://doi.org/10.1007/s00044-025-03495-1
- McFadden, W. M., Snyder, A. A., Kirby, K. A., et al. Rotten to the core: antivirals targeting the HIV-1 capsid core.Retrovirology. 2021;18:41.https://doi.org/10.1186/s12977-021-00583-z
- Xu, S., et al. Twice-yearly lenacapavir demonstrated exceptional efficacy for HIV prevention. The Innovation. 2024;5(5):100683. https://doi.org/10.1016/j.xinn.2024.100683
Disclaimer
The editors take care to share authentic information. In case of any discrepancies please write to newsletter@medness.org.
The sponsors do not have any influence on the nature or kind of the news/analysis reported in MedNess. The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of MedNess. Examples of analysis performed within this article are only examples. They should not be utilized in real-world analytic products as they are based only on very limited and dated open-source information. Assumptions made within the analysis are not reflective of the position of anyone volunteering or working for MedNess. This blog is strictly for news and information. It does not provide medical advice, diagnosis or treatment nor investment suggestions. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or another qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
MedNess is a part of STEMPeers® which is a 501(c)(3) organization registered in PA as PhD Career Support Group. The organization helps create a growing network of STEM scientists that is involved in peer-to-peer mentoring and support.